
#Iso 13485 clauses iso
Although the European Union Directives do not mandate certification to ISO 9001 and/or ISO 13485 the preferred method to prove compliance to such standards is to seek its official certification which is issued by certifying organizations known as 'Registrars'. One of the major requirements to prove conformity is the implementation of the Quality Management System according ISO 9001 and/or ISO 13485 and ISO 14971. The conformity of Medical Devices and In-vitro Diagnostic Medical Device according to European Union Directives 93/42/EEC, 90/385/EEC and 98/79/EEC must be assessed before sale is permitted.
#Iso 13485 clauses verification
specific requirements for verification of the effectiveness of corrective and preventive actionsĬompliance with ISO 13485 is often seen as the first step in achieving compliance with European regulatory requirements.specific requirements for documentation and validation of processes for sterile medical devices.
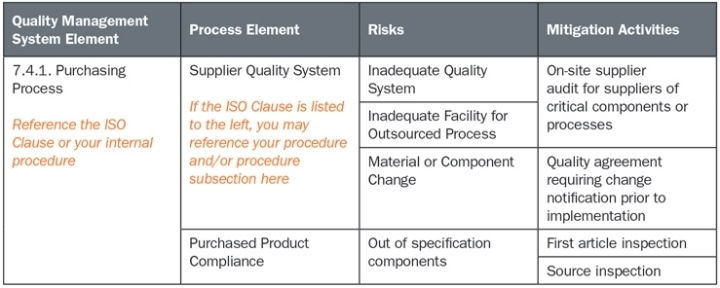
specific requirements for inspection and traceability for implantable devices.focus on risk management activities and design control activities during product development.controls in the work environment to ensure product safety.Food and Drug Administration (FDA), or the Medical Devices Directive 93/42/EEC, required for doing business in the European Union Examples of market-specific regulatory requirements include 21 CFR 820, the Quality System Regulation for medical devices sold in the United States, enforced by the U.S. the promotion and awareness of regulatory requirements as a management responsibility.Additionally, the ISO 9001 requirements regarding customer satisfaction are absent from the medical device standard. A principal difference, however, is that ISO 9001 requires the organization to demonstrate continual improvement, whereas ISO 13485 requires only that the certified organization demonstrate the quality system is effectively implemented and maintained. While it remains a stand-alone document, ISO 13485 is generally harmonized with ISO 9001. Though it is tailored to the industry's quality system expectations and regulatory requirements, an organization does not need to be actively manufacturing medical devices or their components to seek certification to this standard, in contrast to the automotive sector's ISO/TS 16949, where only firms with an active request for quotation, or on the bid list, of an International Automotive Task Force supply chain manufacturer can seek registration. Requirements of ISO are applicable to organizations regardless of their size and regardless of. ISO can also be used by suppliers or external parties that provide product, including quality management system-related services to such organizations. The current ISO 13485 edition was published on 1 March 2016. This standard supersedes earlier documents such as EN 46001 (19) and EN 46002 (1996), the previously published ISO 13485 (19), and ISO 13488 (also 1996). ISO 13485 Medical devices - Quality management systems - Requirements for regulatory purposes is an International Organization for Standardization (ISO) standard published for the first time in 1996 it represents the requirements for a comprehensive quality management system for the design and manufacture of medical devices. ISO 13485 Third edition Reference number ISO (E) Licensed to Red Star Contract Mfg / Barry Leffers ( ) ISO Store Order: OP-125087 / Downloaded: Single user licence only, copying and networking prohibited.Any activity that receives input and converts it to output can be considered as a process. This International Standard is based on a process approach to quality management.
#Iso 13485 clauses pdf
